CAPTACO2 – First results
20 June 2022
CAPTACO2 project, led by PhD student Anna Mas, which is looking for the most efficient way to capture carbon dioxide from the environment using a small-sized capture module, is yielding very interesting data.
Adjusting all the elements that integrate the experiment as detailed in the post “CAPTACO2 – Absorption testing has proved to be a difficult task. Setting up the gas chromatograph was especially complicated because of the control parameters configuration and because of the incident air flow adjustment.
With the experiment running, the data we receive allows us to study how much carbon dioxide stays inside the absorption module over time, and then plot it on a graph so that we can compare different results.
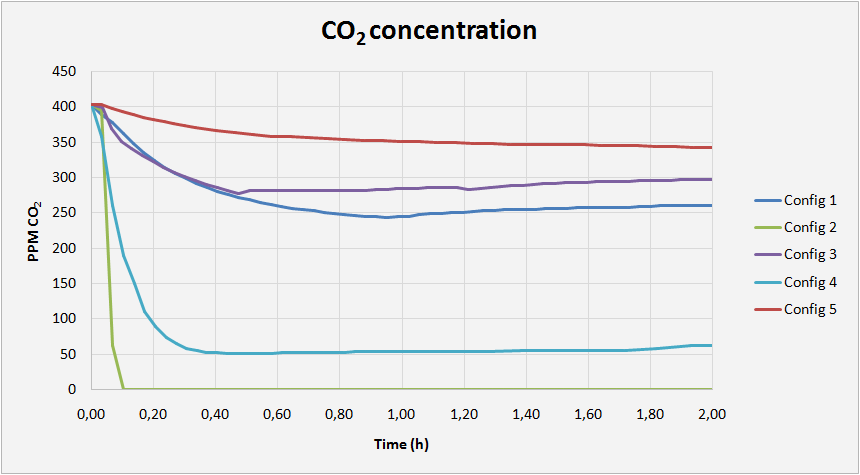
The module in which carbon dioxide is absorbed receives air with a known composition, where CO2 concentration is around 400ppm (similar to atmospheric air), while the gas chromatograph measures CO2 concentration at the module outlet. From this data it is possible to know how much carbon dioxide has been absorbed, and how fast it has been absorbed.
And this is what the graph shows. There are different absorption curves depending on the type of absorbent and the type of membrane used. For example, the green line (Config 2) corresponds to an experiment in which the absorbent is KOH (potassium hydroxide) and there is no separation membrane between the air and the absorbent. These special conditions provide a very fast absorption.
Other configurations do not provide such good results, but all this information together allows us to advance in our research to absorb carbon dioxide as fast as possible in order to fight against climate change.












